 
By the 1920s and 1930s temperatures close to absolute zero were reached, and by 1960 laboratories could produce temperatures of 0.000001 K, a millionth of a degree Kelvin above absolute zero. Three years later the propensity of many supercooled metals to lose all resistance to electricity-the phenomenon known as superconductivity-was discovered. In 1895, when it had become possible to reach temperatures as low as 40 K, air was liquefied and separated into its major components in 1908 helium was liquefied (4.2 K). Since then the theoretical development of cryogenics has been connected to the growth in capability of refrigeration systems. Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers.The word "cryogenic" is Greek and roughly translates to mean "to generate cold." Cryogenics had its beginning in 1877, the year that oxygen was first cooled to the point at which it became a liquid (−183 ☌, 90 K). All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. To say that the temperature is between 24˚C and 26˚C is the same as saying between 75 and 79˚F. It's because the degree size is smaller, and that affords us a little more "dynamic range," or more subtlety in variation between close temperatures. 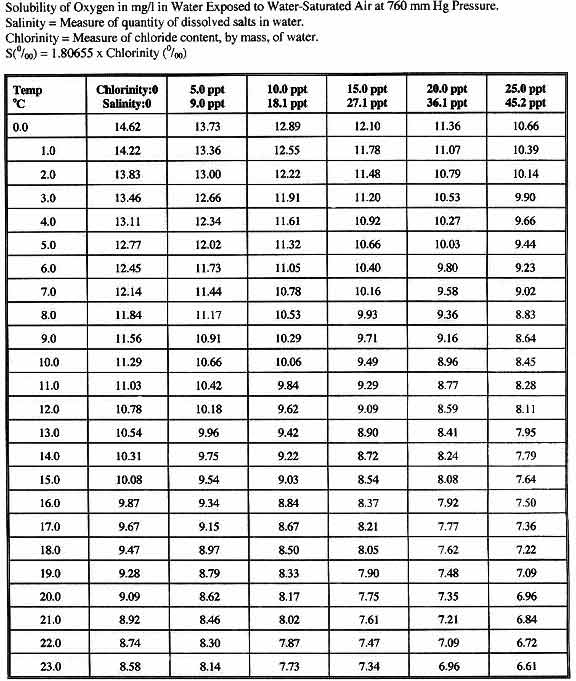
Most would agree that Celsius and Kelvins are the appropriate choice for science, but many feel like, when it comes to describing the temperature outside or in the house, the Fahrenheit system is a little easier. The Celsius degree and the Kelvin (it's not "Kelvin degrees") are the same size.Įven scientists sometimes disagree about which temperature scale to use. The F degree is smaller than the C degree. There are fewer Celsius degrees between these two temperatures than Fahrenheit degrees. Remember that the size of the Celsius degree and the Kelvin are the same. 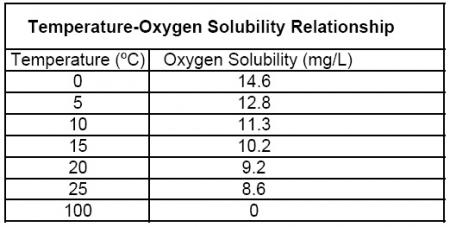
The atoms are, relatively-speaking, closely packed at low temperature.Īs more heat is added (next figure), the average speed of the atoms increases, which makes collisions more energetic, resulting in two phenomena.Ĭonvert 37˚C to both Fahrenheit degrees and Kelvin. This setup insures a constnat pressure because the mass always balances the internal pounding of of the piston by the atoms. Pressure is the avarage force of the impacts of the atoms on the piston (a function of their speed and the frequency with which they strike the piston). The piston is weighted with a mass, which causes a certain pressure inside. In the figure below, gas atoms are enclosed in a container with a movable piston. The figures below illustrate what happens to a gas of atoms as the heat is added to it. We exploit the property of thermal expansion of materials to make devices to measure heat as temperature. It is an imperfect measure of the total heat content of a system because it doesn't directly measure all of the rotational or vibrational energy of molecules, but those can be calculated fairly accurately from the temperature. Temperature is a measure of the heat content of a substance or system. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
